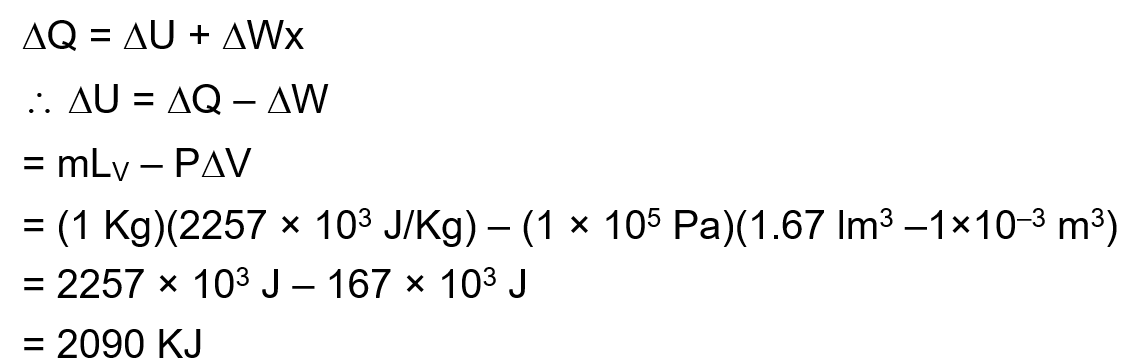1 kg of water at 100°C is converted into steam at 100°C by boiling at atmospheric pressure. The volume of water changes from $1.00 \times 10^{-3} \mathrm{~m}^3$ as a liquid to 1.671 m 3 as steam. The change in internal energy of the system during the process will be (Given latent heat of vaporisaiton = 2257 kJ/kg. Atmospheric pressure $=1 \times 10^5 \mathrm{~Pa}$ )