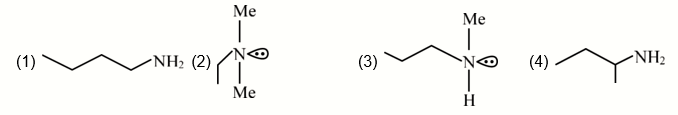An organic compound $[\mathrm{A}]\left(\mathrm{C}_4 \mathrm{H}_{11} \mathrm{~N}\right)$, shows optical activity and gives $\mathrm{N}_2$ gas on treatment with $\mathrm{HNO}_2$. The compound [A] reacts with $\mathrm{PhSO}_2 \mathrm{Cl}$ producing a compound which is soluble in KOH. The structure of A is: