For the reaction, $\mathrm{aA}+\mathrm{bB} \rightarrow \mathrm{cC}+\mathrm{dD}$, the plot of $\log \mathrm{k}$ vs $\frac{1}{\mathrm{~T}}$ is given below :
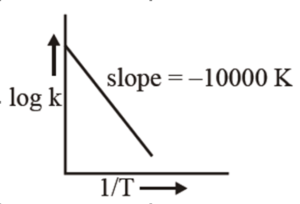
The temperature at which the rate constant of the reaction is $10^{-4} \mathrm{~s}^{-1}$ is $\_\_\_\_$ K.
(Rounded-off to the nearest integer)
[Given The rate constant of the reaction is $10^{-5} \mathrm{~s}^{-1}$ at 500 K .]

