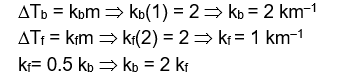Elevation in the boiling point for 1 molal solution of glucose is 2 K . The depression in the freezing point for 2 molal solution of glucose in the same solvent is 2 K . The relation between $\mathrm{K}_{\mathrm{b}}$ and $\mathrm{K}_{\mathrm{f}}$ is :