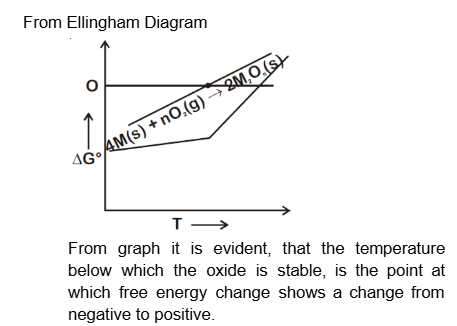
For a reaction,
$$
4 \mathrm{M}(\mathrm{~s})+\mathrm{nO}_2(\mathrm{~g}) \rightarrow 2 \mathrm{M}_2 \mathrm{O}_{\mathrm{n}}(\mathrm{~s})
$$
the free energy change is plotted as a function of temperature. The temperature below which the oxide is stable could be inferred from the plot as the point at which