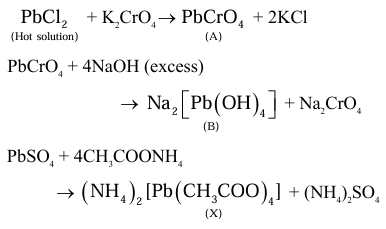Consider the following reactions.
$\mathrm{PbCl}_2+\mathrm{K}_2 \mathrm{CrO}_4 \rightarrow \mathrm{~A}+2 \mathrm{KCl}$ (Hot solution)
$$
\begin{aligned}
& \mathrm{A}+\mathrm{NaOH} \rightleftharpoons \mathrm{~B}+\mathrm{Na}_2 \mathrm{CrO}_4 \\
& \mathrm{PbSO}_4+4 \mathrm{CH}_3 \mathrm{COONH}_4 \rightarrow\left(\mathrm{NH}_4\right)_2 \mathrm{SO}_4+\mathrm{X}
\end{aligned}
$$
In the above reactions, $\mathrm{A}, \mathrm{B}$ and X are respectively.