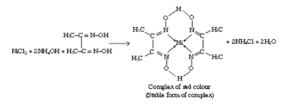
 In the above complex, Ni is present in +2 oxidation number.
A) It is rosy red ppt
B) It is precipitated in basic medium
C) $\mathrm{Ni}^{+2}: 3 \mathrm{~d}^8$
Hybridisation : $\mathrm{dsp}^2$
Unpaired $\mathrm{e}^{-}=0$
Geometry : Square planar
D) $\mathrm{N}-\mathrm{Ni}-\mathrm{N}$ Bond angle is close to $90^{\circ}$
E) 2 five membered metal containing rings are formed.
In the above complex, Ni is present in +2 oxidation number.
A) It is rosy red ppt
B) It is precipitated in basic medium
C) $\mathrm{Ni}^{+2}: 3 \mathrm{~d}^8$
Hybridisation : $\mathrm{dsp}^2$
Unpaired $\mathrm{e}^{-}=0$
Geometry : Square planar
D) $\mathrm{N}-\mathrm{Ni}-\mathrm{N}$ Bond angle is close to $90^{\circ}$
E) 2 five membered metal containing rings are formed.