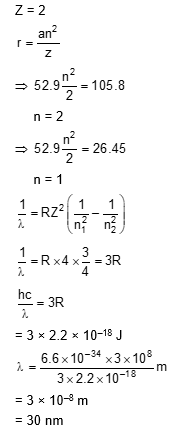For $\mathrm{He}^{+}$, a transition takes place from the orbit of radius 105.8 pm to the orbit of radius 26.45 pm . The wavelength (in nm ) of the emitted photon during the transition is $\_\_\_\_$ .
[Use:
Bohr radius, $\mathrm{a}=52.9 \mathrm{pm}$
Rydberg constant, $\mathrm{RH}_{\mathrm{H}}=2.2 \times 10^{-18} \mathrm{~J}$
Planck's constant, $\mathrm{h}=6.6 \times 10^{-34} \mathrm{~J} \mathrm{~s}$
Speed of light, $\mathrm{c}=3 \times 10^8 \mathrm{~m} \mathrm{~s}^{-1}$ ]