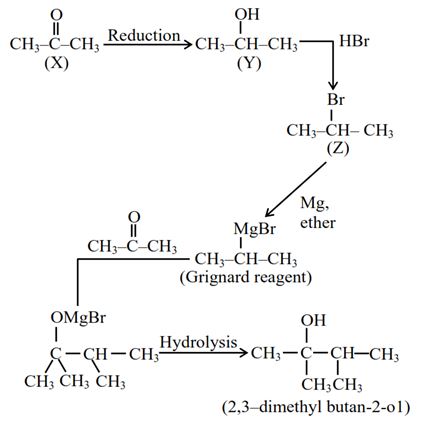
An organic compound (X) with molecular formula ${{\rm{C}}_3}{{\rm{H}}_6}{\rm{O}}$ is not readily oxidised. On reduction it gives ${{\rm{C}}_3}{{\rm{H}}_8}{\rm{O}}({\rm{Y}})$ which reacts with HBr to give a bromide (Z) which is converted to Grignard reagent. This Grignard reagent on reaction with (X) followed by hydrolysis give 2,3-dimethylbutan-2-ol. Compounds (X),(Y) and (Z) respectively are: