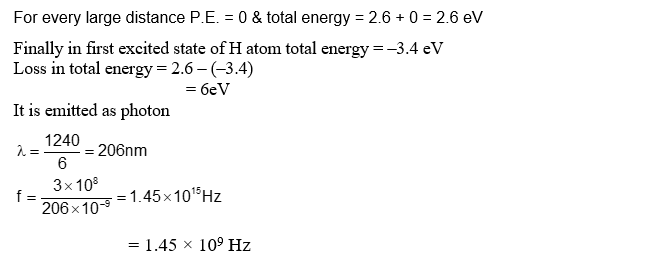A free electron of 2.6 eV energy collides with a $\mathrm{H}_{+}$ion. This results in the formation of a hydrogen atom in the first excited state and a photon is released. Find the frequency of the emitted photon. $\left(\mathrm{h}=6.6 \times 10^{-34} \mathrm{Js}\right)$