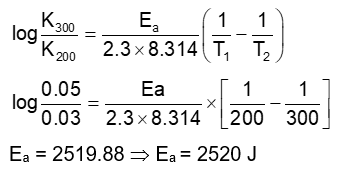$$
A \rightarrow B
$$
The rate constants of the above reaction at 200 K and 300 K are $0.03 \mathrm{~min}^{-1}$ and $0.05 \mathrm{~min}^{-1}$ respectively. The activation energy for the reaction is $\_\_\_\_$ J (Nearest integer)
(Given : In $10=2.3, \mathrm{R}=8.3 \mathrm{JK}^{-1} \mathrm{~mol}^{-1}, \log 5=0.70, \log 3=0.48, \log 2=0.30$ )