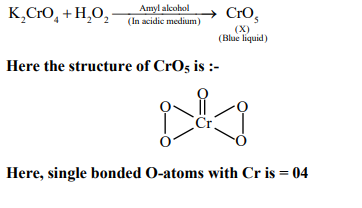
An acidified solution of potassium chromate was layered with an equal volume of amyl alcohol. When it was shaken after the addition of 1 mL of $3 \% \mathrm{H}_2 \mathrm{O}_2$, a blue alcohol layer was obtained. The blue color is due to the formation of a chromium (VI) compound ' $\mathbf{X}$ '. What is the number of oxygen atoms bonded to chromium through only single bonds in a molecule of $\mathbf{X}$ ?