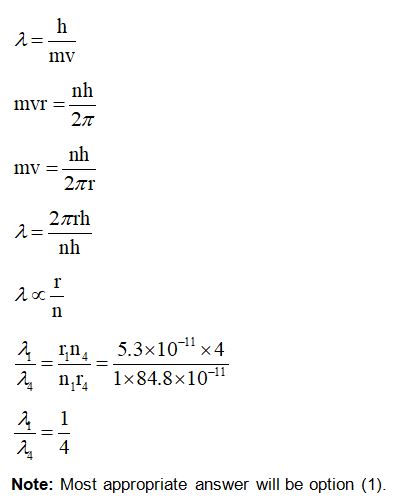An electron in the ground state of the hydrogen atom has the orbital radius of $5.3 \times {10^{ - 11}}\;{\rm{m}}$ while that for the electron in third excited state is $8.48 \times {10^{ - 10}}\;{\rm{m}}$ . The ratio of the de Broglie wavelengths of electron in the ground state to that in the excited state is