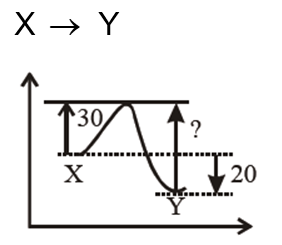
An exothermic reaction $X \rightarrow Y$ has an activation energy $30 \mathrm{~kJ} \mathrm{~mol}^{-1}$. If energy change $\Delta \mathrm{E}$ during the reaction is -20 kJ , then the activation energy for the reverse reaction in kJ is $\_\_\_\_$ .(Integer answer)