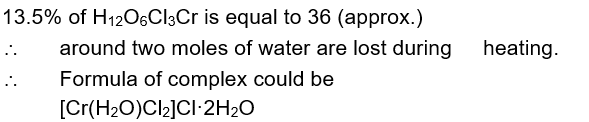Complex A has a composition of $\mathrm{H}_{12} \mathrm{O}_6 \mathrm{Cl}_3 \mathrm{Cr}$. If the complex on treatment with conc. $\mathrm{H}_2 \mathrm{SO}_4$ loses $13.5 \%$ of its original mass, the correct molecular formula of A is [Given : atomic mass of $\mathrm{Cr}=52$ amu and $\mathrm{Cl}=35 \mathrm{amu}$ ]