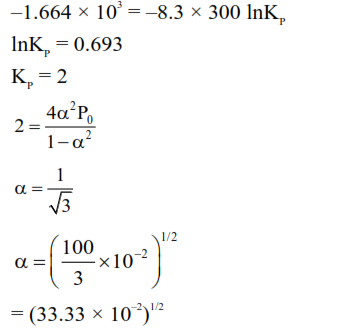Dissociation of a gas $\mathrm{A}_2$ takes place according to the following chemical reaction. At equilibrium, the total pressure is 1 bar at 300 K .
$\mathrm{A}_2(\mathrm{~g}) \rightleftharpoons 2 \mathrm{~A}(\mathrm{~g})$
The standard Gibbs energy of formation of the involved substances has been provided below:
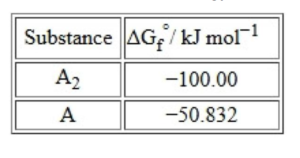
The degree of dissociation of $\mathrm{A}_2(\mathrm{~g})$ is given by $\left(x \times 10^{-2}\right)^{1 / 2}$ where $x=$ $\_\_\_\_$ . (Nearest integer).
[Given: $\mathrm{R}=8 \mathrm{~J} \mathrm{~mol}^{-1} \mathrm{~K}^{-1}, \log 2=0.3010, \log 3=0.48$ ]
Assume degree of dissociation is not negligible.