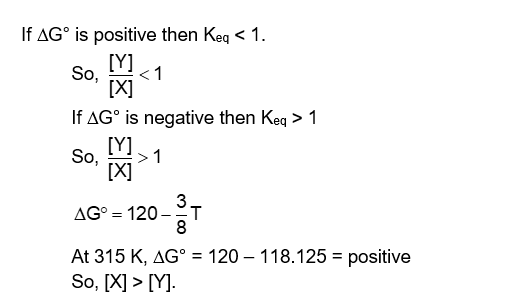For the chemical reaction $\mathrm{X} \rightleftharpoons \mathrm{Y}$, the standard reaction Gibbs energy depends on temperature T (in K ) as $\Delta_{\mathrm{r}} \mathrm{G}^{\circ}\left(\right.$ in $\left.\mathrm{kJ} \mathrm{mol}^{-1}\right)=120-\frac{3}{8} \mathrm{~T}$. The major component of the reaction mixture at T is