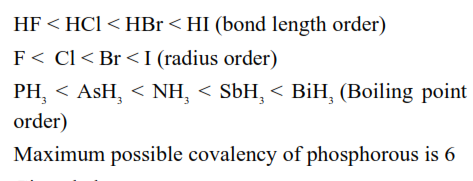
Given below are two statements:
Statement I: The halogen that makes longest bond with hydrogen in HX, has the smallest covalent radius in its group.
Statement II: A group 15 element's hydride $\mathrm{EH}_3$ has the lowest boiling point among corresponding hydrides of other group 15 elements. The maximum covalency of that element E is 4 .
In the light of the above statements, choose the correct answer from the options given below