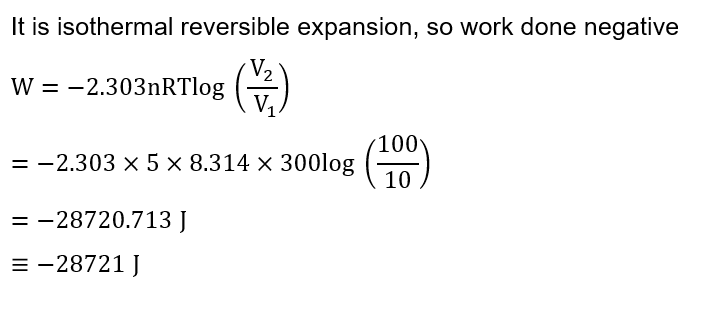If 5 moles of an ideal gas expands from 10 L to a volume of 100 L at 300 K under isothermal and reversible condition then work, w , is $-x \mathrm{~J}$. The value of $x$ is $\_\_\_\_$
(Given $\mathrm{R}=8.314 \mathrm{~J} \mathrm{~K}^{-1} \mathrm{~mol}^{-1}$ )