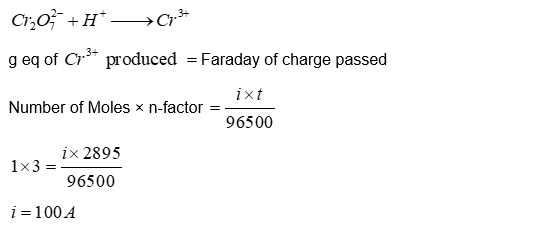In an electrochemical cell, dichromate ions in aqueous acidic medium are reduced to $\mathrm{Cr}^{3+}$. The current (in amperes) that flows through the cell for 48.25 minutes to produce 1 mole of $\mathrm{Cr}^{3+}$ is $\_\_\_\_$ .
Use: 1 Faraday $=96500 \mathrm{Cmol}^{-1}$