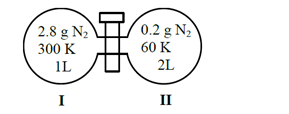
Two flasks I and II shown below are connected by a valve of negligible volume.
When the valve is opened, the final pressure of the system in bar is $x \times 10^{-2}$. The value of $x$ is $\_\_\_\_$ .
(Integer answer)
[Assume-Ideal gas; 1 bar $=10^5 \mathrm{~Pa}$; Molar mass of $\mathrm{N}_2=28.0 \mathrm{~g} \mathrm{~mol}^{-1} ; \mathrm{R}=8.31 \mathrm{~J} \mathrm{~mol}^{-1} \mathrm{~K}^{-1}$ ]