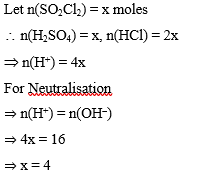$\mathrm{SO}_2 \mathrm{Cl}_2$ on reaction with excess of water results into acidic mixture
$$
\mathrm{SO}_2 \mathrm{Cl}_2+2 \mathrm{H}_2 \mathrm{O} \rightarrow \mathrm{H}_2 \mathrm{SO}_4+2 \mathrm{HCl}
$$
16 moles of NaOH is required for the complete neutralisation of the resultant acidic mixture. The number of moles of $\mathrm{SO}_2 \mathrm{Cl}_2$ used is :