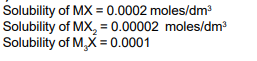Solubility product constants $\left(\mathrm{K}_{s p}\right)$ of salts of types $\mathrm{MX}, \mathrm{MX}_2$ and $\mathrm{M}_3 \mathrm{X}$ at temperature " T " are $4.0 \times 10^{-8}, 3.2 \times 10^{-14}$ and $2.7 \times 10^{-15}$, respectively. Solubilities $\left(\mathrm{mol} \mathrm{dm}^{-3}\right)$ of the salts at temperature " $T$ " are in the order