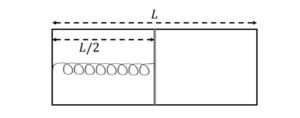
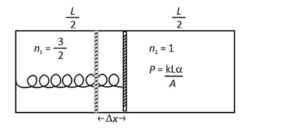
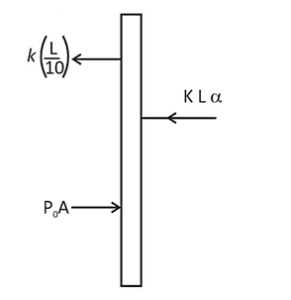
The left and right compartments of a thermally isolated container of length $L$ are separated by a thermally conducting, movable piston of area $A$. The left and right compartments are filled with $\frac{3}{2}$ and 1 moles of an ideal gas, respectively. In the left compartment the piston is attached by a spring with spring constant $k$ and natural length $\frac{2 L}{5}$. In thermodynamic equilibrium, the piston is at a distance $\frac{L}{2}$ from the left and right edges of the container as shown in the figure. Under the above conditions, if the pressure in the right compartment is $P=\frac{k L}{A} \alpha$, then the value of $\alpha$ is $\_\_\_\_$ .