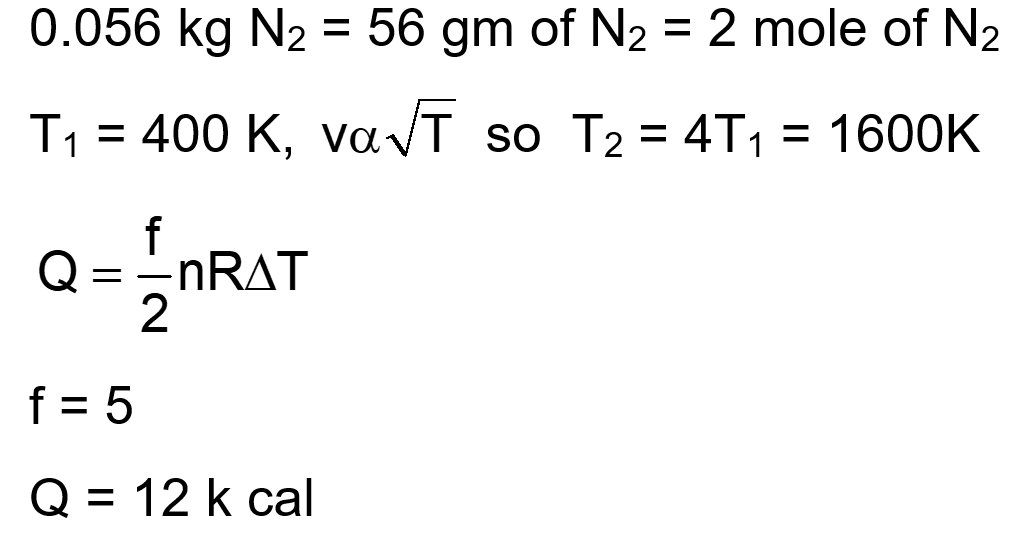0.056 kg of Nitrogen is enclosed in a vessel at a temperature of $127^{\circ} \mathrm{C}$. The amount of heat required to double the speed of its molecules is $\_\_\_\_$ $\mathrm{k} \mathrm{cal} .\left(\right.$ Take $\mathrm{R}=2$ cal mole $\left.^{-1} \mathrm{~K}^{-1}\right)$