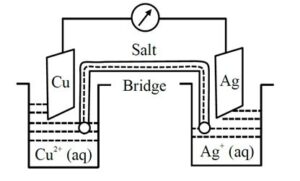
1 Faraday electricity was passed through ${\rm{C}}{{\rm{u}}^{2 + }}(1.5{\rm{M}},1\;{\rm{L}})/{\rm{Cu}}$ and 0.1 Faraday was passed through ${\rm{A}}{{\rm{g}}^ + }(0.2{\rm{M}},1\;{\rm{L}})/{\rm{Ag}}$ electrolytic cells. After this the two cells were connected as shown below to make an electrochemical cell. The emf of the cell thus formed at 298 K is____mV (nearest integer)
Given : ${{\rm{E}}^\circ }{\rm{C}}{{\rm{u}}^{2 + }}/{\rm{Cu}} = 0.34\;{\rm{V}}$
${{\rm{E}}^\circ }{\rm{A}}{{\rm{g}}^ + }/{\rm{Ag}} = 0.8\;{\rm{V}}$
$\frac{{2 \cdot 303{\rm{RT}}}}{{\rm{F}}} = 0.06\;{\rm{V}}$
 Given : ${{\rm{E}}^\circ }{\rm{C}}{{\rm{u}}^{2 + }}/{\rm{Cu}} = 0.34\;{\rm{V}}$
Given : ${{\rm{E}}^\circ }{\rm{C}}{{\rm{u}}^{2 + }}/{\rm{Cu}} = 0.34\;{\rm{V}}$