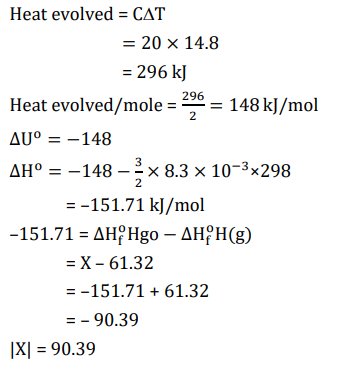2 mol of $\mathrm{Hg}(\mathrm{g})$ is combusted in a fixed volume bomb calorimeter with excess of $\mathrm{O}_2$ at 298 K and 1 atm into $\mathrm{HgO}(\mathrm{s})$. During the reaction, temperature increases from 298.0 K to 312.8 K . If heat capacity of the bomb calorimeter and enthalpy of formation of $\mathrm{Hg}(\mathrm{g})$ are $20.00 \mathrm{~kJ} \mathrm{~K}^{-1}$ and 61.32 $\mathrm{kJ} \mathrm{mol}^{-1}$ at 298 K , respectively, the calculated standard molar enthalpy of formation of $\mathrm{HgO}(\mathrm{s})$ at $298 \mathrm{~K} \mathrm{is}^{-1} \mathrm{X} \mathrm{kJ} \mathrm{mol}^{-1}$. The value of $|\mathrm{X}|$ is $\_\_\_\_$ .
[Given: Gas constant $\mathrm{R}=8.3 \mathrm{~J} \mathrm{~K}-1 \mathrm{~mol}-1$ ]