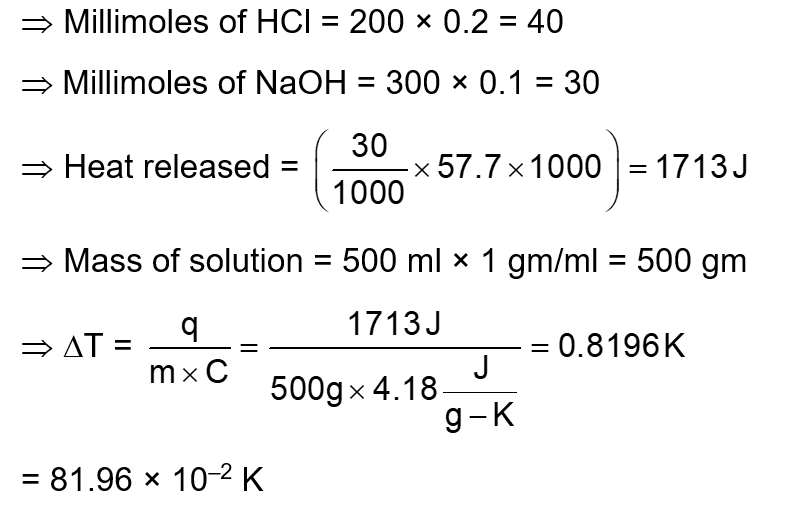
200 mL of 0.2 M HCl is mixed with 300 mL of 0.1 M NaOH . The molar heat of neutralization of this reaction is -57.1 kJ . The increase in temperature in ${ }^{\circ} \mathrm{C}$ of the system on mixing is $x \times 10^{-2}$.
The value of $x$ is $\_\_\_\_$ (Nearest integer)
[Given : Specific heat of water $=4.18 \mathrm{~J} \mathrm{~g}^{-1} \mathrm{~K}^{-1}$
Density of water $=1.00 \mathrm{~g} \mathrm{~cm}^{-3}$ ]
(Assume no volume change on mixing)