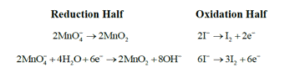
Adding oxidation half and reduction half, net reaction is
$$
\begin{aligned}
& 2 \mathrm{MnO}_4^{-}+6 \mathrm{I}^{-}+4 \mathrm{H}_2 \mathrm{O} \rightarrow 3 \mathrm{I}_2+2 \mathrm{MnO}_2+8 \mathrm{OH}^{-} \\
& \Rightarrow \mathrm{z}=8 \\
& \Rightarrow \text { Ans } 8
\end{aligned}
$$