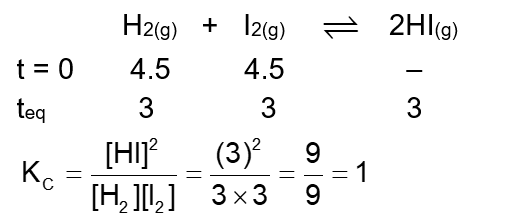4.5 moles each of hydrogen and iodine is heated in a sealed ten litre vessel. At equilibrium, 3 moles of HI were found. The equilibrium constant for $\mathrm{H}_2(\mathrm{~g})+\mathrm{I}_2(\mathrm{~g}) \rightleftharpoons 2 \mathrm{HI}(\mathrm{g})$ is $\_\_\_\_$ .