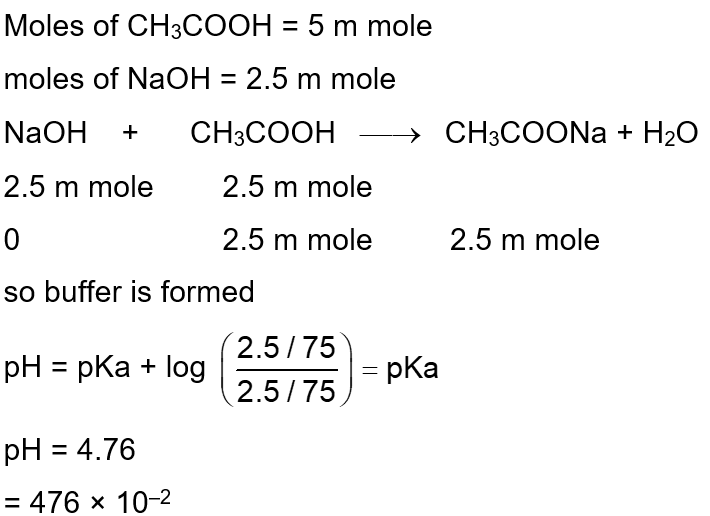
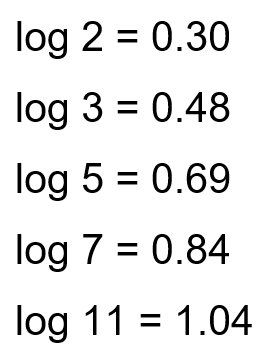50 mL of $0.1 \mathrm{M} \mathrm{CH}_3 \mathrm{COOH}$ is being titrated against 0.1 M NaOH . When 25 mL of NaOH has been added, the pH of the solution will be $\_\_\_\_$ $\times 10^{-2}$. (Nearest integer)
(Given : $\left.\mathrm{pKa}\left(\mathrm{CH}_3 \mathrm{COOH}\right)=4.76\right)$