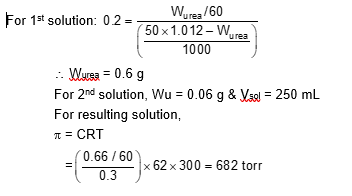
50 mL of 0.2 molal urea solution (density $=1.012 \mathrm{~g} \mathrm{~mL}^{-1}$ at 300 K ) is mixed with 250 mL of a solution containing 0.06 g of urea. Both the solutions were prepared in the same solvent. The osmotic pressure (in Torr) of the resulting solution at 300 K is $\_\_\_\_$ .
[Use:
Molar mass of urea $=60 \mathrm{~g} \mathrm{~mol}^{-1}$; gas constant, $\mathrm{R}=62 \mathrm{~L} \mathrm{Torr} \mathrm{K}^{-1} \mathrm{~mol}^{-1} \xlongequal{\text {. }}$ Assume, $\Delta$ mix $\mathrm{H}=0, \Delta$ mix $\mathrm{V}=0$ ]