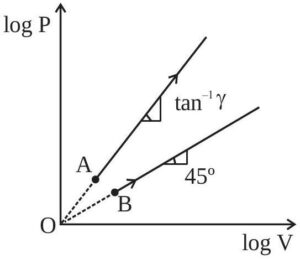
Two thermodynamical process are shown in the figure. The molar heat capacity for process A and B are $C_A$ and $C_B$. The molar heat capacity at constant pressure and constant volume are represented by $C_P$ and $C_V$, respectively. Choose the correct statement.