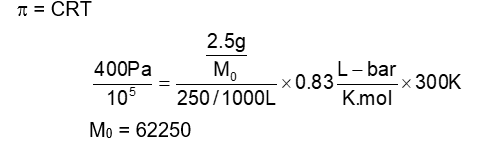
At $27^{\circ} \mathrm{C}$, a solution containing 2.5 g of solute in 250.0 mL of solution exerts an osmotic pressure of 400 Pa . The molar mass of the solute is $\_\_\_\_$ $\mathrm{g} \mathrm{mol}^{-1}$ (Nearest integer)
(Given : $\mathrm{R}=0.083 \mathrm{~L} \mathrm{bar} \mathrm{K}^{-1} \mathrm{~mol}^{-1}$ )