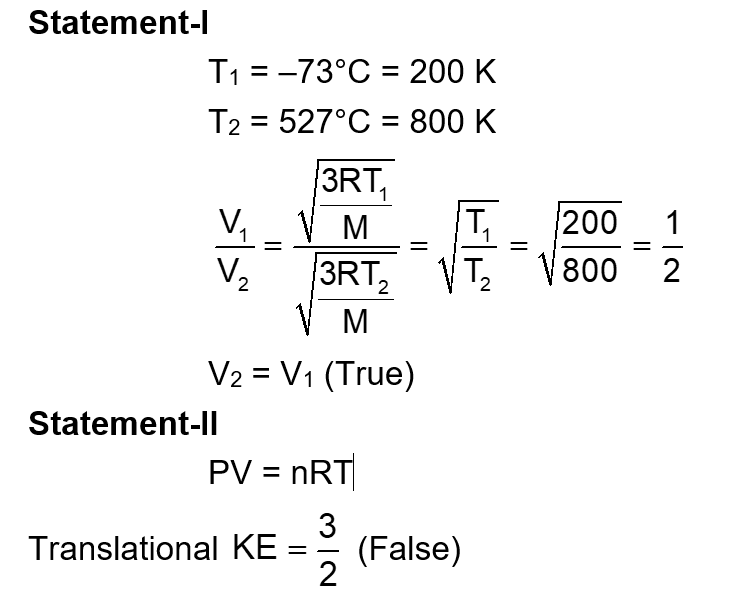
Given below are two statements :
Statements 1 : The temperature of a gas is $-73^{\circ} \mathrm{C}$. When the gas is heated to $527^{\circ} \mathrm{C}$, the root mean square speed of the molecules is doubled.
Statement II: The product of pressure and volume of an ideal gas will be equal to translational kinetic energy of the molecules.
In the light of the above statements, choose the correct answer from the options given below :