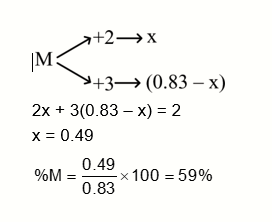A sample of a metal oxide has formula $\mathrm{M}_{0.83} \mathrm{O}_{100}$. The metal M can exist in two oxidation states +2 and +3. In the sample of $\mathrm{M}_{0.83} \mathrm{O}_{100}$, the percentage of metal ions existing in +2 oxidation state is _____% (nearest integer)