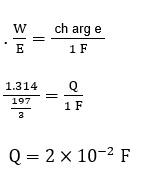A constant current was passed through a solution of $\mathrm{AuCl}_4^{-}$ion between gold electrodes. After a period 10.0 minutes, the increase in mass of cathode was 1.314 g . The total charge passed through the solution is $\times 10^{-2} \mathrm{~F}$.
(Given atomic mass of $\overline{\mathrm{Au}}=197$ )