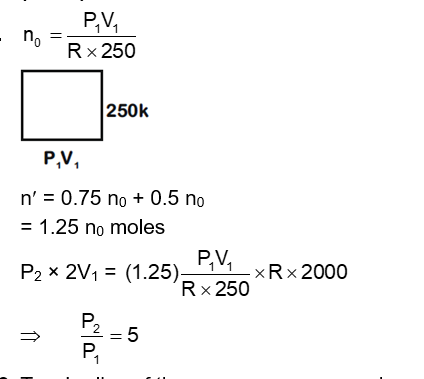
Initially a gas of diatomic molecules is contained in a cylinder of volume $\mathrm{V}_1$ at a pressure $\mathrm{P}_1$ and temperature 250 K . Assuming that $25 \%$ of the molecules get dissociated causing a change in number of moles. The pressure of the resulting gas at temperature 2000 K , when contained in a volume $2 V_1$ is given by $P_2$. The ratio $P_2 / P_1$ is