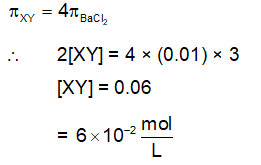The osmotic pressure of a dilute solution of an ionic compound XY in water is four times that of a solution of 0.01 M $\mathrm{BaCl}_2$ in water. Assuming complete dissociation of the given ionic compounds in water, the concentration of XY (in mol $\mathrm{L}^{-1}$) in solution is