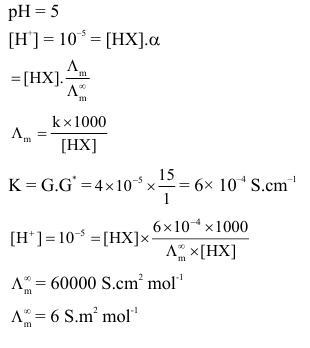The pH and conductance of a weak acid $(\mathrm{HX})$ was found to be 5 and $4 \times 10^{-5} \mathrm{~S}$, respectively. The conductance was measured under standard condition using a cell where the electrode plates having a surface area of $1 \mathrm{~cm}^2$ were at a distance of 15 cm apart. The value of the limiting molar conductivity is $\_\_\_\_$ $\mathrm{Sm}^2 \mathrm{~mol}^{-1}$. (nearest integer)
(Given : degree of dissociation of the weak acid $(\alpha) \ll 1$ )