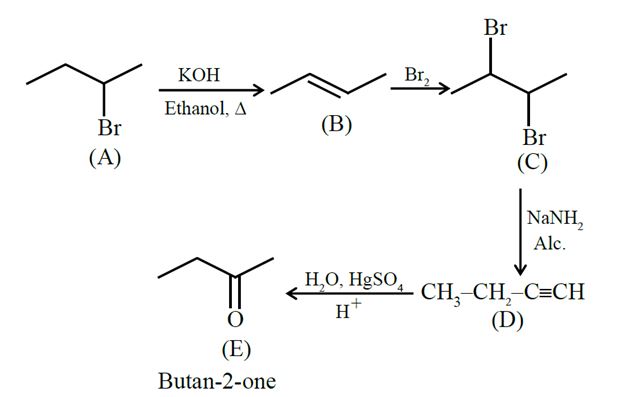
An optically active alkyl halide ${C_4}{H_9}Br[A]$ reacts with hot KOH dissolved in ethanol and forms alkene [B] as major product which reacts with bromine to give dibromide [C]. The compound [C] is converted into a gas [D] upon reacting with alcoholic $NaN{H_2}$. During hydration 18 gram of water is added to 1 mole of gas [D] on warming with mercuric sulphate and dilute acid at 333 K to form compound [E]. The IUPAC name of compound [E] is :