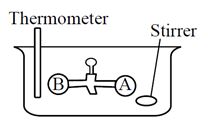
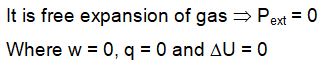 Two vessels A and B are connected via stopcock. The vessel A is filled with a gas at a certain pressure. The entire assembly is immersed in water and is allowed to come to thermal equilibrium with water. After opening the stopcock the gas from vessel A expands into vessel B and no change in temperature is observed in the thermometer. Which of the following statement is true ?
Two vessels A and B are connected via stopcock. The vessel A is filled with a gas at a certain pressure. The entire assembly is immersed in water and is allowed to come to thermal equilibrium with water. After opening the stopcock the gas from vessel A expands into vessel B and no change in temperature is observed in the thermometer. Which of the following statement is true ?