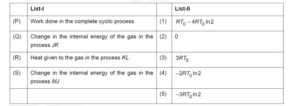
One mole of a monatomic ideal gas undergoes the cyclic process $J \rightarrow K \rightarrow L \rightarrow M \rightarrow J$, as shown in the $P-T$ diagram.
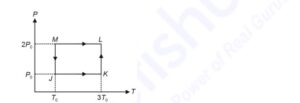
Match the quantities mentioned in List-I with their values in List-II and choose the correct option.
[R is the gas constant.]