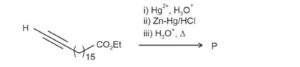
In the following reaction sequence, the major product P is formed .
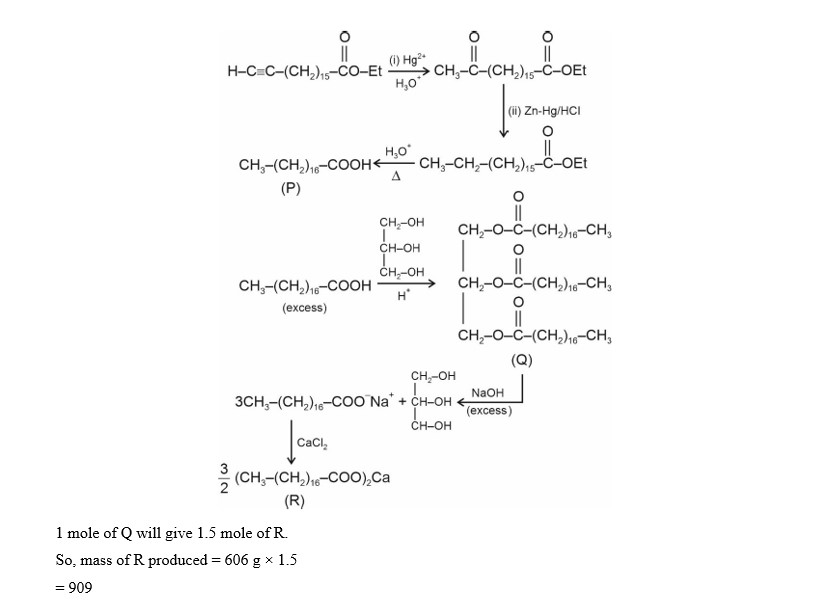
Glycerol reacts completely with excess $\mathbf{P}$ in the presence of an acid catalyst to form $\mathbf{Q}$. Reaction of $\mathbf{Q}$ with excess NaOH followed by the treatment with $\mathrm{CaCl}_2$ yields Ca -soap $R$, quantitatively. Starting with one mole of $Q$, the amount of $\mathbf{\mathbf { R }}$ produced in gram is
[Given, atomic weight: $\mathrm{H}=1, \mathrm{C}=12, \mathrm{~N}=14, \mathrm{O}=16, \mathrm{Na}=23, \mathrm{Cl}=35, \mathrm{Ca}=40$ ]
 Glycerol reacts completely with excess $\mathbf{P}$ in the presence of an acid catalyst to form $\mathbf{Q}$. Reaction of $\mathbf{Q}$ with excess NaOH followed by the treatment with $\mathrm{CaCl}_2$ yields Ca -soap $R$, quantitatively. Starting with one mole of $Q$, the amount of $\mathbf{\mathbf { R }}$ produced in gram is
Glycerol reacts completely with excess $\mathbf{P}$ in the presence of an acid catalyst to form $\mathbf{Q}$. Reaction of $\mathbf{Q}$ with excess NaOH followed by the treatment with $\mathrm{CaCl}_2$ yields Ca -soap $R$, quantitatively. Starting with one mole of $Q$, the amount of $\mathbf{\mathbf { R }}$ produced in gram is