In a conductometric titration, small volume of titrant of higher concentration is added stepwise to a larger volume of titrate of much lower concentration, and the conductance is measured after each addition.
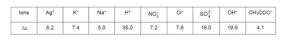
The limiting ionic conductivity $\left(\Lambda_0\right)$ values (in $\mathrm{mSm}^2 \mathrm{~mol}^{-1}$ ) for different ions in aqueous solutions are given below:
For different combinations of titrates and titrants given in List-I, the graphs of ‘conductance’ versus ‘volume of titrant’ are given in List-II.
Match each entry in List-I with the appropriate entry in List-II and choose the correct option.


