The plot of $\log k_f$ versus $1 / \mathrm{T}$ for a reversible reaction $\mathrm{A}(\mathrm{g}) \rightleftharpoons \mathrm{P}(\mathrm{g})$ is shown.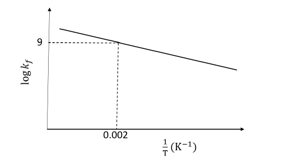Pre-exponential factors for the forward and backward reactions are $10^{15} \mathrm{~s}^{-1}$ and $10^{11} \mathrm{~s}^{-1}$, respectively. If the value of $\log K$ for the reaction at 500 K is 6 , the value of $\left|\log k_b\right|$ at 250 K is $\_\_\_\_$
[ $\mathrm{K}=$ equilibrium constant of the reaction
$k_f=$ rate constant of forward reaction
$k_b=$ rate constant of backward reaction]
