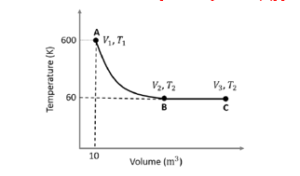
 $A \rightarrow B$ is an adiabatic process. If the total heat absorbed in the entire process $(A \rightarrow B$ and $B \rightarrow C)$ is $R T_2$ In 10 , the value of $2 \log V_3$ is $\_\_\_\_$ -
[Use, molar heat capacity of the gas at constant pressure, $C_{B M}=\frac{5}{2} R$ ]
$A \rightarrow B$ is an adiabatic process. If the total heat absorbed in the entire process $(A \rightarrow B$ and $B \rightarrow C)$ is $R T_2$ In 10 , the value of $2 \log V_3$ is $\_\_\_\_$ -
[Use, molar heat capacity of the gas at constant pressure, $C_{B M}=\frac{5}{2} R$ ]