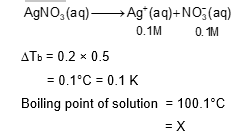The boiling point of water in a 0.1 molal silver nitrate solution (solution A ) is $\mathrm{x}^{\circ} \mathrm{C}$. To this solution A , an equal volume of 0.1 molal aqueous barium chloride solution is added to make a new solution $B$. The difference in the boiling points of water in the two solutions A and B is $\mathrm{y} \times 10^{-2}{ }^{\circ} \mathrm{C}$.
(Assume: Densities of the solutions A and B are the same as that of water and the soluble salts dissociate completely.
Use: Molal elevation constant (Ebullioscopic Constant), $\mathrm{K}_{\mathrm{b}}=0.5 \mathrm{~K} \mathrm{~kg} \mathrm{~mol}^{-1}$; Boiling point of pure water as $100^{\circ} \mathrm{C}$.)
The value of $x$ is $\_\_\_\_$ .