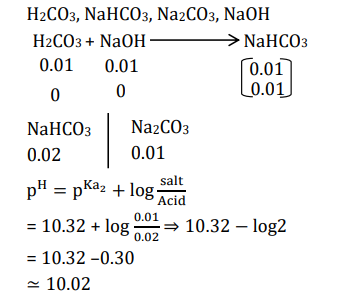A solution is prepared by mixing 0.01 mol each of $\mathrm{H}_2 \mathrm{CO}_3, \mathrm{NaHCO}_3, \mathrm{Na}_2 \mathrm{CO}_3$, and NaOH in 100 mL of water. pH of the resulting solution is $\_\_\_\_$ .
[Given: $\mathrm{pK}_1$ and $\mathrm{pK}_2$ of $\mathrm{H}_2 \mathrm{CO}_3$ are 6.37 and 10.32, respectively; $\log 2=0.30$ ]