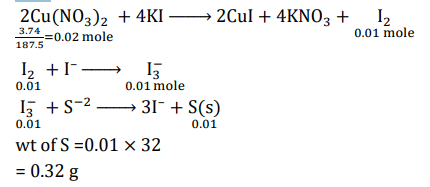The treatment of an aqueous solution of 3.74 g of $\mathrm{Cu}\left(\mathrm{NO}_3\right)_2$ with excess KI results in a brown solution along with the formation of a precipitate. Passing $\mathrm{H}_2 \mathrm{~S}$ through this brown solution gives another precipitate $\mathbf{X}$. The amount of $\mathbf{X}$ (in g ) is $\_\_\_\_$ .
[Given: Atomic mass of $\mathrm{H}=1, \mathrm{~N}=14, \mathrm{O}=16, \mathrm{~S}=32, \mathrm{~K}=39, \mathrm{Cu}=63, \mathrm{I}=127$ ]